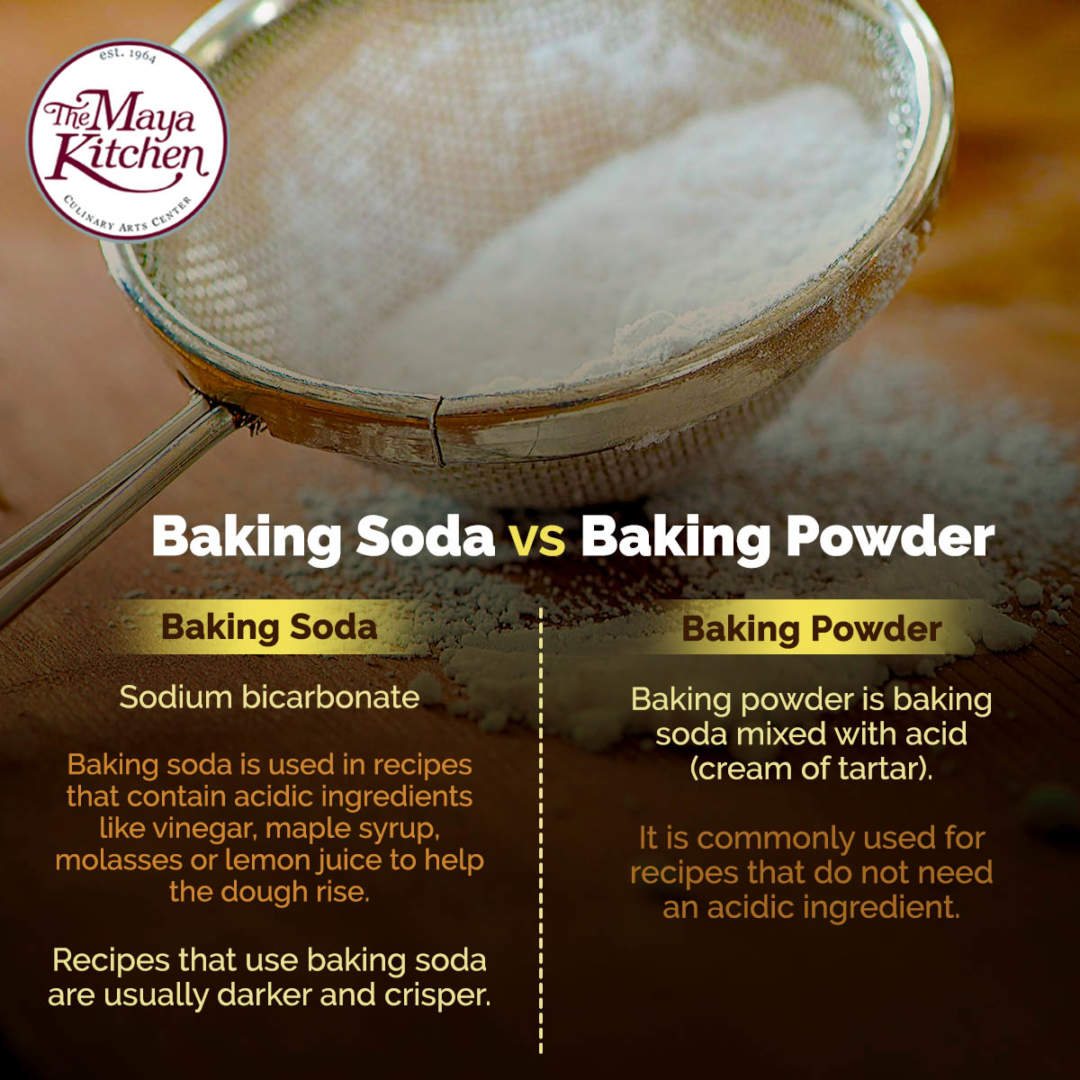
…it can affect both browning and flavour.– to add more lift., creating a lighter, fluffier and well risen baked product. brown sugar, yoghurt etc.), however the carbon dioxide created from the acid and baking soda is not enough to leaven the volume of the batter in the recipe. recipes that require both baking powder and baking soda also contain an acid (e.g.…sometimes we need more leavening than we have acid available in the recipe. Why do some recipes call for both baking soda and baking powder? Since baking powder already contains an acid, it is more often used when a recipe does not call for an additional acidic ingredient.Ī good rule of thumb is 1 teaspoon to 125g (1 cup) of flour. This is called double acting baking powder and this is the most common type of baking powder.ģ. In just 1 teaspoon of baking powder there is 1/4 teaspoon of baking soda – I told you baking soda was strong!īaking powder will begin reacting when it is wet but will do most of the reacting once it is heated. Baking powder is a mixture of baking soda (base) and cream of tartar (acid), therefore it is already neutralised.īaking powder also contains cornstarch which works to separate the acid and base as well as keeping them both dry by absorbing moisture. always use baking soda with an acidic ingredient for optimal taste and riseġ.Thus, getting the right amount of baking soda in your baking is important, we don’t want soapy tasting cakes! Remember also that too much baking soda in a recipe and not enough acid will still result in a soapy, metallic taste in the baked good. the less baking soda) the more easily it will set. For example, the more acidic the recipe (i.e. Baking soda is strong – about 3-4 times stronger than baking powder, so you don’t need much.ġ/4 teaspoon to 125g (1 cup) flour is usually the rule of thumb when baking, however it also depends on how much acid is in your recipe and what texture you would like. This is why sometimes you cannot prepare batters ahead of time to bake later as the baking soda has already done a lot of the reacting process.Ģ. when exposed to heat), it’s best to bake the batter immediately for optimal rise.

Even though carbon dioxide will continue to be produced in the oven (i.e. Do note that because baking soda reacts when mixed with an acid, it’s important to get the cake batter into the oven straight away. When it is neutralised, carbon dioxide is released faster which removes the soapy taste.Īcidic ingredients include: lemon juice, yoghurt, vinegar, honey, cocoa, brown sugar, applesauce, molasses, buttermilk (to name a few). This is because baking soda is a base and needs an acid to neutralise it. However: when baking soda is heated without an acid, carbon dioxide is released slowly which leaves a yucky soapy, metallic tasting cake.